Will a Water Bottle Explode in the Freezer? Yes!
Yes, a water bottle can explode in the freezer. When water freezes, it expands, and if there is not enough space in the bottle for this expansion, the pressure can cause the bottle to burst.
Water has a unique property in that it expands when it transitions from a liquid to a solid state. This occurs due to the molecular structure of ice, which takes up more space than liquid water.
The expansion can exert pressure on the container holding the water. In the case of a sealed water bottle, the rigid sides may not be able to accommodate the expanding ice, leading to a rupture or explosion.
To prevent water bottles from exploding in the freezer, leave some space at the top of the bottle before freezing, or use a freezer-safe bottle designed to handle expansion.
Key Takeaway
The Freezing Process and Its Effects

The freezing process can cause liquid water to expand and potentially exert significant pressure on the container.
When a water bottle is placed in the freezer, the drop in temperature can lead to the formation of ice, which occupies a greater volume than its liquid counterpart.
As a result, the expansion of the water as it freezes can put stress on the walls of the container. This pressure may cause the bottle to deform or, in extreme cases, rupture.
Understanding the physical properties of water and the implications of its freezing process is essential in designing containers that can withstand such pressures.
Innovations in materials and container design can mitigate the risk of explosion, ensuring the safety and integrity of water bottles when exposed to freezing temperatures.
Understanding Water Expansion
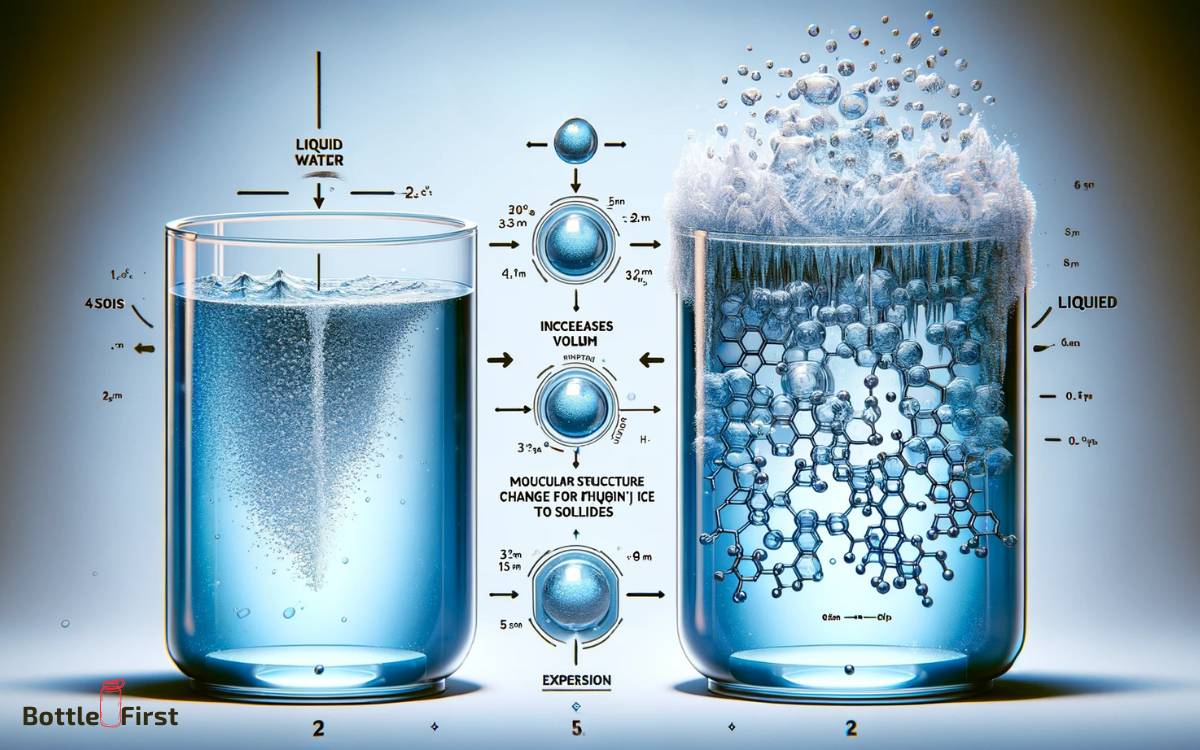
Upon freezing, water undergoes a phase transition, resulting in an increase in volume and pressure within the container, which can lead to deformation or rupture.
This phenomenon is a result of water expanding as it freezes, unlike most other substances that contract when transitioning from a liquid to a solid state.
The expansion occurs due to the formation of crystalline structures in the water molecules, causing them to arrange themselves in a way that takes up more space than when they were in a liquid state.
As a result, the pressure inside a sealed container can build up significantly, potentially causing the container to burst.
Understanding this expansion is crucial in assessing the risks associated with freezing a sealed container, which will be further explored in the following section.
Risks of Freezing a Sealed Container

When freezing water in a sealed container, the increased volume and pressure resulting from water expansion can pose the risk of deformation or rupture.
As water freezes, it expands by approximately 9%, exerting immense pressure on the container. This pressure can cause plastic bottles to warp, glass bottles to crack, or aluminum cans to burst.
The risk is amplified if the container is overfilled, as there is inadequate space for the water to expand.
Moreover, the structural integrity of the container and the material it is made of significantly influence the likelihood of deformation or rupture.
Therefore, it is crucial to use containers specifically designed for freezing liquids, ensuring they have sufficient room for water expansion and are made from materials capable of withstanding the associated pressures.
Factors That Influence Freezing Explosions

Factors influencing freezing explosions include the container’s material, size, and the presence of air pockets. The material of the container determines its ability to expand without rupturing.
For instance, plastic containers are more flexible than glass, reducing the risk of explosion. Additionally, the size of the container affects the rate of freezing. Smaller volumes freeze faster, creating higher internal pressure.
Lastly, air pockets within the liquid can create weak points, increasing the likelihood of an explosion.
Understanding these factors is crucial for designing containers that can withstand freezing temperatures without compromising safety.
| Factor | Influence | Example |
|---|---|---|
| Material | The presence of air pockets within the liquid can lead to explosions | Plastic containers are more flexible than glass |
| Size | Affects the rate of freezing, creating higher internal pressure | Smaller volumes freeze faster, increasing internal pressure |
| Air pockets | Create weak points, increasing the likelihood of an explosion | Presence of air pockets within the liquid can lead to explosions |
Preventing Freezing Mishaps
When it comes to preventing freezing mishaps, there are several key points to consider.
First, it’s important to avoid filling the water bottle completely to leave room for expansion as the liquid freezes.
Additionally, using plastic bottles instead of glass can help prevent potential explosions.
Avoid Filling Completely
To prevent freezing mishaps, moderation in filling water bottles is essential. Avoiding overfilling the bottles allows room for expansion when the water freezes, preventing the container from bursting.
It is important to leave some space at the top of the bottle to accommodate the expansion of the freezing water.
The following table provides a guideline for filling water bottles to prevent them from bursting in the freezer:
| Bottle Size | Recommended Fill Level |
|---|---|
| 16 oz | 14 oz |
| 32 oz | 28 oz |
| 64 oz | 56 oz |
| 1 gallon | 7/8 full |
By following these recommendations, you can avoid the potential hazards of overfilling water bottles and ensure a safe freezing process.
Leave Room for Expansion
Leaving sufficient space in a water bottle to accommodate the expansion of freezing water is crucial for preventing the bottle from bursting in the freezer.
To ensure a successful freezing process, consider the following innovative tips:
- Use a bottle with a wide mouth to allow for better expansion.
- Fill the bottle only about 3/4 full to leave ample room for the water to expand as it freezes.
Consider using silicone ice trays to freeze water instead of bottles.
- Place the bottle on its side in the freezer to maximize surface area and allow for more even expansion.
- Invest in a specialized freezer-safe water bottle designed to withstand the expansion of freezing liquids.
Use Plastic, Not Glass
Plastic bottles are preferred over glass for freezing water due to their ability to flex and expand, reducing the risk of bursting in the freezer.
When selecting a container for freezing water, it is crucial to consider the material to prevent potential mishaps.
Plastic bottles offer the flexibility needed to accommodate the expansion of water as it freezes, safeguarding against breakage.
To emphasize the advantages of using plastic over glass for freezing water, consider the following comparison table:
| Aspect | Plastic Bottles | Glass Bottles |
|---|---|---|
| Flexibility | Able to flex and expand without breaking | Brittle and prone to cracking under freezing pressure |
| Safety | Less likely to shatter if the water expands too much | High risk of shattering when water expands |
| Convenience | Lightweight and easy to handle | Heavier and more cumbersome |
Safe Thawing Practices

Thaw frozen water bottles slowly and consistently to avoid potential ruptures or damage.
When it comes to safely thawing frozen water bottles, consider the following innovative practices:
- Place the frozen water bottle in the refrigerator and allow it to thaw slowly over several hours.
- Submerge the frozen water bottle in a bowl of cold water, replacing the water every 30 minutes until thawed.
- Use a microwave to thaw the water bottle, but be cautious and follow the microwave’s instructions to prevent overheating.
- Utilize a thawing tray specifically designed to safely thaw frozen items at room temperature.
- Consider investing in a portable electronic thawing device that uses controlled heat to thaw frozen water bottles safely and efficiently.
Conclusion
It is important to understand the potential dangers of freezing a sealed water bottle.
The expansion of water as it freezes can cause the bottle to explode, resulting in a messy and potentially dangerous situation.
By taking precautions and considering the factors that influence freezing explosions, such as the type of bottle and temperature, one can avoid this unfortunate outcome.
Remember, a frozen water bottle may not be as refreshing as you think!






